The PICASO project ended 30 June 2019 and a final newsletter is now available. We also invite you to visit our Knowledge centre where you can watch the two PICASO videos: “Sharing care plans” and “Care efficiency, conflict identification and patient control”. In the Knowledge centre you will also find a list of all the public deliverables, newsletters, presentations, publications and the final PICASO brochure which highlights the project’s key achievements.
PICASO video – Care efficiency, conflict identification and patient control of data
Watch how the PICASO self-monitoring solution supports patient empowerment for patients with multimorbidities – in this case a woman suffering from rheumatoid arthritis and cardiovascular conditions. She is actively engaged by measuring health parameters at home and sharing this data with her physicians and informal caregivers, who can identify possible treatment overlap and optimise treatment.
PICASO innovation in the final for the Innovation Radar Prize 2018
Project partner CNet has been selected as one of 50 finalists for the European Commission‘s Innovation Radar Prize 2018 with the PICASO innovation: Federated Cloud Architecture designed for ehealth: Care Management as a Service.
The Innovation Radar Prize is a prestigious prize that puts a spotlight on high quality innovative excellence, emerging from the Horizon 2020 programme. Finalists are grouped into five categories, with 10 finalists in each category, and CNet has been selected for the “Tech for Society” category, recognising technologies impacting society and citizens.
The innovation
Care Management as a Service is a cloud solution that enables care organisations to use cloud technology and experience benefits of scalability and software resource sharing without violating regulations regarding clinical data. The innovation is based on a software-to-data cloud approach. All software for care management and decision support is hosted in one public cloud while all clinical data always resides inside the care organisations. All access and use of clinical data are done in a secure and authorised way, without violating GDPR.
“This is an innovation that healthcare has been looking for since cloud technology was introduced. We are very proud to have been selected and it is an important recognition of the innovative work being carried out in the PICASO project”, says Peter Rosengren, technical coordinator of PICASO and CEO of CNet.
Vote for your favourite
Thousands of EU-funded innovations from across Europe were scanned in the process, making it quite an achievement to make the final 50. But it is not over yet. The public has to vote for the favourite innovators, who will then pitch their innovation in front of a jury before a winner can be announced.
Vote for CNet’s innovation HERE

Join our workshop on e-health solutions for home hospitalisation and patient empowerment
PICASO and the POLYCARE project invite you to a discussion of e-health solutions and their effect on the provision of healthcare services in terms of cost and sustainability. The event takes place on 14th November 2018 in Sankt Augustin, Germany.
Care plan management using PICASO
The PICASO platform helps to overcome the core obstacles to a continuum of care which include: complexity of managing large amounts of data, absence of essential patient data and limited collaboration between different clinical domains. It does so by providing healthcare professionals with a comprehensive overview of a patient’s situation and data through a web-based interface and by enabling the creation and sharing of care plans.
The solution combines clinical, home monitoring and risk assessment data about a patient and gives access to all shared data about that patient. It consists of four main management tools: Patient Data Viewer, Data Resource Browser, Care Plan Manager and Risk Manager.
All relevant data presented
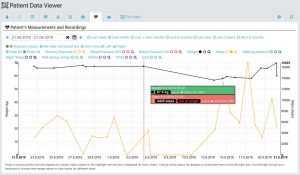
The Patient Data Viewer shows all relevant data about the patient, including basic information and comments, medication and treatment history, visits and appointments.
It displays home monitoring measurements and recordings, enabling comparison over time, and a medication adherence score, identifying any non-adherence with cardiovascular medications. Finally, it provides an overview of diseases that the patient suffers from and appointed clinicians.

Search for shared patient information
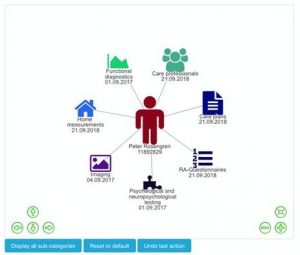
To view shared information about a patient, the healthcare professional can do a search in the Data Resource Browser, yielding a dynamic, interactive mind map of all data that the healthcare professional is allowed to access. The mind map is shown as a graph where the healthcare professional can click on the individual elements to see the associated information if authorised to do so.
If, for example, a general practitioner (GP) searches for data sent by a patient, it is possible to see all the carers who have requested data, and which data the carer has received from the patient. Additionally, the GP can click on a certain data type and see all the measurements performed and also see other carers’ interventions and the care plans executed.
Creating and sharing the care plan
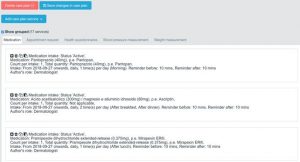
In the Care Plan Manager, the healthcare professional can create, integrate and share a patient’s care plan between all involved professional carers. The care plans are stored as templates in a narrative form whereby certain services can be added.
In case of medication issues, the healthcare professional is notified by the system and can adjust treatment accordingly.
Assessing risk
To support decision making, the healthcare professional can use the Risk Manager to calculate risk scores based on patient information. For the trials, basic cardiovascular risk scores are used. It is also possible to simulate results based on change of input, e.g. predicting the effect if a patient stops smoking or manages to lower the cholesterol levels.
The system is also able to generate health alerts based on the patient data, either to patients or clinicians. However, this is currently not being implemented due to the implications in terms of responsibility. Alerts sent to patients might also trigger concern so it has to be implemented with great care.
Related article: Positive feedback from the clinical trials
Positive feedback from the clinical trials
The PICASO ICT platform is now being tested in two different clinical and national settings. Preliminary results show that PICASO facilitates the management of patients and medication adherence.
Two main aspects of the PICASO platform are tested in the trials: the sharing of patient data and care plans across care sectors for a more holistic treatment of people with multimorbidities. And the involvement of patients in their own care for a better health outcome.
The trials
One trial is carried out in Rome, Italy, by the University Tor Vergata and the Department of Psychiatry of the Institute of Treatment and Research, Santa Lucia of Rome. It involves 30 patients over 65 years of age with Parkinson’s Disease and an associated comorbidity. Ten patients will use PICASO and 20 will be part of a control group. Health professionals using the PICASO platform include: radiologists, nuclear medicine physicians, neurologists, psychiatrists and neuropsychologists.
The other trial takes place in Germany, organised by the Policlinic of Rheumatology & Hiller Research Unit at the Heinrich-Heine University in Düsseldorf. Here, the target is 30 patients who attend the outpatient clinic. They are over 18 years of age and have Rheumatoid Arthritis with a cardiovascular comorbidity. The patients will evaluate the platform together with their physicians (rheumatologists, rheumatologists in training and others) over a period of 6 months.
Sharing data and care plans
The two clinical trials use the PICASO ICT platform to support a collaborative sharing of patient data and care plans between the different health professionals involved in the treatment. The clinician user interface shows the overall picture of the patient and enables the clinician to adjust care.
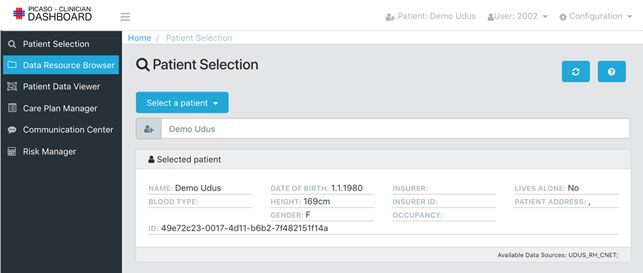
In Italy where the first PICASO patient was enrolled in March 2018, the system is running successfully:
– From a clinical perspective, both physicians and healthcare professionals are very positive so far. They have access to both the clinical history and the home monitoring data, and this facilitates the management of their patients, says Dr. Agostino Chiaravalloti from the University Hospital of Tor Vergata, who leads the trial in Italy.
The PICASO functionalities have also generated several positive expectations about the future interaction among physicians:
– The system allows direct and easy contact between different physicians, who will be able to manage several aspects and problems that derive from daily clinical activities. This saves time and causes less discomfort for patients, says Dr. Agostino Chiaravalloti.
Involve patients in their own care
Enrolled patients are equipped with a home monitoring solution whereby they can measure relevant physiological parameters at home and submit selected observations to the hospital/clinic. This empowers them to play a more active part in their own care and to influence treatment.
The patients can view their measurements and status through a user interface which also gives them an overview of planned activities with status indications and a reminder function, the latter of which is particularly useful:
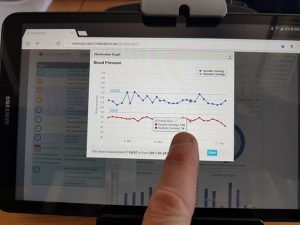
– There is a general consensus among the Italian patients that the patient dashboard provides several benefits in the management of drug intake by means of reminders, says Dr. Agostino Chiaravalloti.
The patient can also grant access to carers such as relatives, nurses or home carers who can access the patient dashboard from a web client.
– Preliminary feedback shows that PICASO seems to help carers in their daily management of patients with chronic conditions. In particular, caregivers are able to check drug intake and other relevant parameters measured by the home monitoring devices. This relieves them from some of their tasks, thus having more time for other clinical duties, explains Dr. Agostino Chiaravalloti.
Secure exchange of patient information
To safely share patient information among all relevant stakeholders, PICASO uses a unique, trust federated solution. The platform operates with divisions into private clouds (care system and patient) and a public cloud for integration purposes. Each trial has a private care cloud where clinical data reside inside the hospital/clinic, and access and use is done by authorization and access restrictions.
Access to patient data is granted based on the written informed consent by the patient, and data are collected, passed on and stored in a pseudonymised way.
High interest in Germany
Whereas the Italian trial has lasted a few months, the German trial has just started and to date, 22 patients have been recruited, and eight physicians are using the PICASO platform. Even though the trial is at an early stage, expectations are high, not only from patients and physicians but also from external stakeholders:
– Both patients and physicians are positive towards the project, but we also experience high interest from other clinical disciplines, patients’ self-help groups and suppliers of health care insurance who we continually are in dialogue with, says Professor Dr. Jutta Richter from the Policlinic of Rheumatology and Hiller Research Unit at the University Hospital of Düsseldorf, who leads the German trial.
The trials will last until February 2019.
Empowering the patient
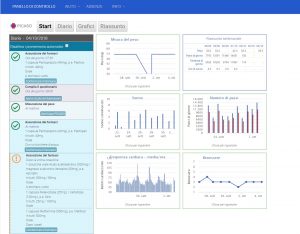
Patients who participate in the two PICASO trials are testing and evaluating the PICASO home monitoring solution.
Home or self-monitoring allows patients a more active role in their own care which in previous studies has shown to have clinical benefits as well as positive effects on chronic patients’ quality of life.
PICASO helps patients to take a more active role in managing their condition by providing an easy-to-use means of measuring and monitoring important health parameters and sharing the data with the relevant clinicians, and in some cases their informal caregivers.
Using a standard tablet, the patient has a dashboard consisting of a care plan diary complete with schedules for medication intake, what health parameters to measure, clinical appointments, etc.
Integrate multiple care plans
An exciting, planned feature of the PICASO care plan diary is the integration of the multiple care plans the patients have if they suffer from multiple morbidities. It makes it simpler for the patient to get an overview of and to adhere to their care plan. PICASO can help them become more in control of their condition, to rely less on their physician for monitoring of specific health parameters and to share their monitored data with all relevant stakeholders by a simple click.
Two video user guides instruct the patient in taking measurements.
Data are only shared with those clinicians who have been granted access by the patients. Behind the “simple click” to share data lies a unique authorisation and security infrastructure which ensures the patients control of their health data.
Understanding the effect of patient empowerment
Better overview, insight and knowledge, more control, and more self-efficacy are essential elements of patient empowerment, and empowered patients are likely to be more engaged in managing their chronic conditions, thus resulting in a better clinical outcome and higher quality of life.
The participants in the PICASO trial will help the project in evaluating how well PICASO facilitates and supports patient empowerment. Although the PICASO trials are small in scale, we expect the results to contribute to a better and uniform understanding of both patient empowerment as a concept, ways of measuring it, and its effect on the health outcome.
Related article: Positive feedback from the clinical trials
PICASO at DGRH 2018
The PICASO platform stood the test at the German Society for Rheumatology 46th Congress 2018 which took place from 19th to 22nd September.
Both the clinician dashboard and the patient dashboard were presented successfully at the exhibition, yielding acknowledgement especially by rheumatologists who considered the solutions to be good and valuable for improving integrated care of their patients.
Apart from interesting suggestions for new features and improvements, physicians also raised a few issues which need to be considered for the exploitability of the solutions.
Over time, there will be a lot of patient information available which the physician has to review since he or she is responsible for treatment, e.g., ensuring that a prescription written by a colleague is not harmful for the patient. This can be time consuming and time is already a problem.
As a result of this issue, IT-platforms like PICASO need to provide features that can serve as valuable clinical decision support to minimise the effort of having to click through a lot of patient information. This means that only relevant information is presented or highlighted, e.g., a recent surgery or a change in medication related to the physician’s speciality.

The EU commission’s risky choice for a non-risk based strategy on assessment of medical devices
PICASO will deploy a home-monitoring solution where the involved patients will be asked to use and test at least three different devices to monitor specific health parameters. The devices used in the project have been carefully selected to ensure that they fulfill the necessary security and quality requirements in order to protect patients, and clinicians, in the trials from harm.
Paul Quinn, representing the project’s legal expert partner, VUB, has published an article in Computer Law & Security Review that discusses the regulation of mHealth devices in the context of the EU Medical Device Framework (MDF) and the how the concept “intended purpose” is used to determine if a device or mobile app have to comply with the MDF. The article entitled “The EU commission’s risky choice for a non-risk based strategy on assessment of medical devices” questions if this key concept is really adequate to protect patient safety.
Article abstract
Regulation of medical devices has been one of the most notable regulatory initiatives of the European Union. The need to ensure that medical devices are of a high quality is self-evident in nature. This is demonstrated by the lack of willingness of both healthcare institutions and professionals to use medical devices that have not properly been certified. In determining which devices are medical devices and should therefore meet the requirements of the regulatory framework, both the current and the proposed frameworks foresee a central place for the concept of ‘intended purpose’. This means that only those manufacturers that have explicitly stated that their device is to be used for a medical purpose should have to comply with the medical device framework. Unfortunately, however, this concept has become increasingly problematic given the rise in mHealth (mobile health) practices and ‘appification’ (shift to mobile devices) in particular, arguably posing potentially serious risks to human health in certain cases. This article discusses the problems that are created by the ever-increasing amount of ‘well-being’ apps and the fact that most will not be classed as medical devices. Despite apparently being aware of these problems, the EU Commission has opted to maintain its current approach in the newly proposed regulation, choosing not to employ other approaches as the FDA has for example done in opting to use a ‘risk based case-by-case approach’.
The Computer Law and Security Review (CLSR) is an international journal of technology law and practice providing a major platform for publication of high quality research, policy and legal analysis within the field of IT law and computer security. The article is available online at www.sciencedirect.com.
PICASO at CareWare 2017
PICASO was represented at the annual CareWare conference which focused on new technologies targeted at the fields of healthcare and assisted living.
The aim with the CareWare Conference was to enhance innovation and the development of technological solutions and the dissemination of knowledge about these technologies and their use. The event took place in Aarhus, Denmark from 4-5th April 2017, and PICASO was part of the exhibition Freedom, Care and Safety.
Read more about the conference here.